|
Since the latter is strong, it also implies that charged particles are accelerated by it, finally settling into a configuration with hardly any remnant electrical force. The gravitational force is only attractive, whereas the electrical force can be attractive or repulsive. This numerical comparison reveals how weak the gravitational force is compared to its electromagnetic counterpart.Īlthough both the forces are inverse-square laws, there is a difference. It is enormous, about 4 followed by 42 zeroes. Instead, the ratio of the electrical and gravitational forces depends on the value of the natural constants, particularly the gravitational constant, the permittivity of vacuum, the electron's charge, and the electron's mass. Since both are inverse square law forces, the distance gets canceled when the ratio of the two forces is considered. Since electrons are fundamental particles, they are a good example. To compare the numerical strengths of the first two, take two particles of the same kind. There are four fundamental forces in nature: the gravitational force, the electromagnetic force, the strong nuclear force, and the weak nuclear force. 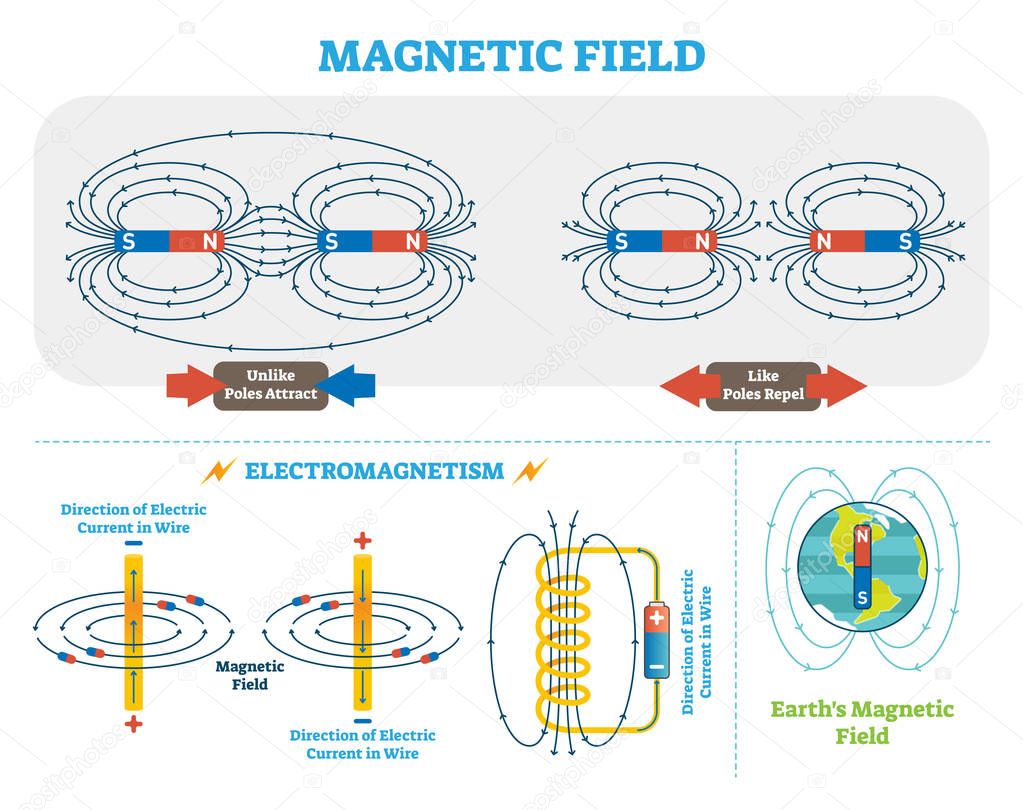
So, it extends over astronomical distances. If there is an electrical force, charged particles move and neutralize it, making objects around us electrically neutral.Īlthough the gravitational force is weak, it cannot be neutralized. For example, a rocket is propelled by electrical forces against the pull of gravity. This enormous number implies that electrical forces are significantly stronger than gravitational forces. 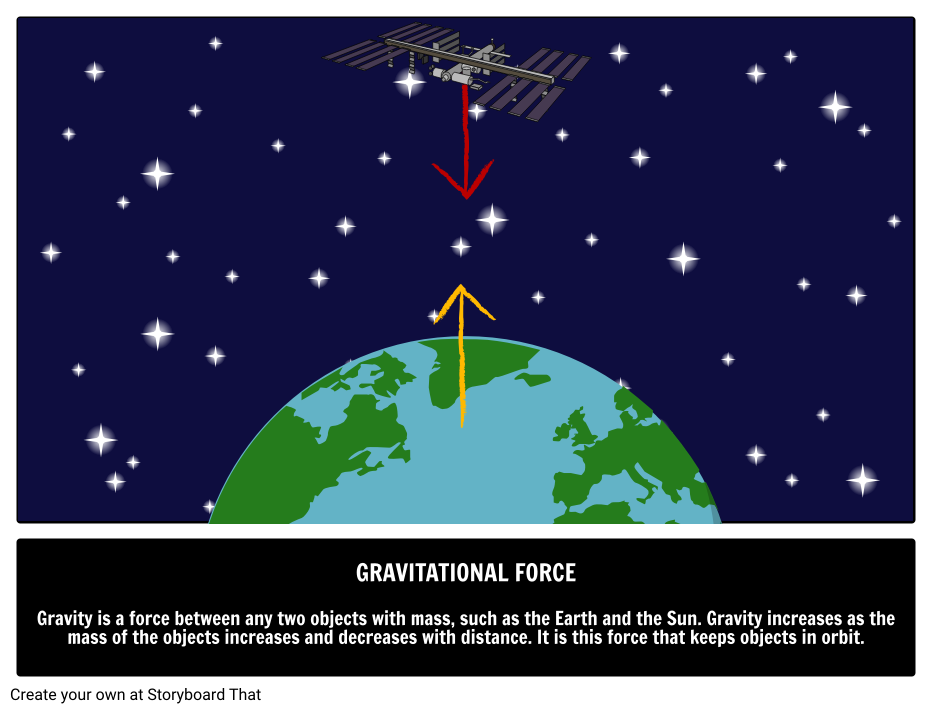
On substitution of their numerical values in SI units, it turns out to be of the order of 10 42. The ratio depends on universal constants and the electron's mass and charge. 
When the magnitudes are compared, the distance dependence cancels. Take two electrons and consider the gravitational and electrostatic forces they exert on each other. Gravitational interaction depends on the product of the point masses, whereas Coulomb interaction depends on the product of the point charges. Whereas gravitational interaction is always attractive, Coulomb interaction may be attractive or repulsive depending on the signs of the electric charges. The Coulomb and the gravitational interactions both depend inversely on the square of the distance between the point objects.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
